The first fuel discovered by humans was likely wood. Subsequently, various animal fats and oils were put to use by our ancestors. Since then, man has discovered many other carbon-based fuels, including coal, petroleum, alcohol, and natural gas. These products contain significant amounts of carbon, which, when burned, produces carbon dioxide (CO2) and other combustion by-products.
Today, there are many times more people on the earth than there were a thousand years ago, more industries than a hundred years ago, and exponentially more motor vehicles of every type than there were just decades ago. Even our electricity demand has never been as high as it is today. What do all these things have in common? Carbon-based fuels directly or indirectly power industries, vehicles, and many other human activities. You could say that the 20th century was the century of carbon, or perhaps more accurately, the petroleum-fueled century.
Green House Gases
The detrimental effects of emissions from all of these carbon-based energy sources became very obvious by the late 1960s. The Environmental Protection Agency (EPA) was created, and environmental legislation was passed mandating vehicles with improved fuel economy and lower tailpipe emissions. The best possible combustion, it was thought at the time, would produce almost no hydrocarbon compounds (HC), very few NOX (nitrogen oxides), zero carbon monoxide (CO), and mostly emit only carbon dioxide (CO2) and water (H2O). CO2 was considered a good gas back then, not a potentially toxic gas like its close relative, carbon monoxide. In the decades since the 1970s, our perspective on CO2 has changed. Today it is considered to be a real problem because it is one of the top-five greenhouse gases.
What is a greenhouse gas? It is a gas or a vapor found in our atmosphere that "absorbs and emits radiant energy within the thermal infrared range, causing the greenhouse effect." In other words, greenhouse gases form a blanket around the earth, capturing and trapping the sun's radiant energy and holding it in our atmosphere, thus raising our planet's temperature. The amount of CO2 in our atmosphere today is the highest it has ever been!
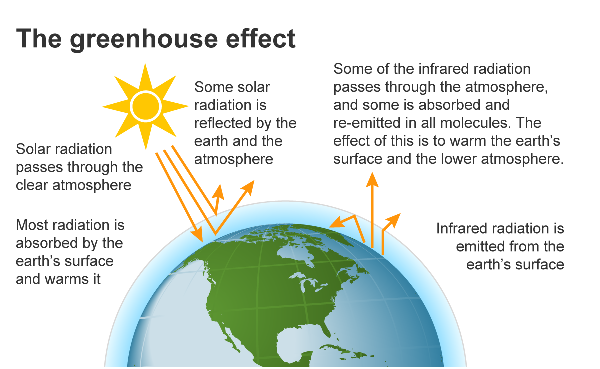
Global Warming is a concept that gained widespread attention several years ago. While many scientists backed this theory, others rejected it. Today, the language has morphed, and 'Climate Change' has largely replaced 'Global Warming.' If you have lived in the United States for the past several years, you will agree that our weather has changed. Many scientists believe we are very close to the tipping point where an increase of even a single degree in the earth's average temperature could set off an irreversible series of catastrophes that would change our geography and lives forever.
Rather than debate which of these theories is most likely to be correct, I suggest we focus on what we can do to avoid the problem. We should focus on doing what we can to keep our planet sustainable and inhabitable. We can agree that carbon dioxide is one of the top-five greenhouse gases. We can agree that it contributes to making the earth warmer. It is the only one of the top-five greenhouse gases which result from the burning of carbon-based fossil fuels. Carbon dioxide is the only greenhouse gas that we can truly control, so it stands to reason that we must work to control and reduce it.
In a concerted effort to limit CO2 emissions, governments worldwide have passed regulations restricting CO2 outputs of vehicles, which essentially require manufacturers to produce electric cars. While these regulations are well-intentioned, they do not necessarily solve the problem. Instead, they switch the emissions from the car's tailpipe to the electrical power plant's smokestack. We cannot produce enough electricity here in the US to power our current demands without using coal, oil, or natural gas.
Here Comes Hydrogen
The increased electrical demand for charging an extensive fleet of battery-powered automobiles will mean power plants will have to burn even more fossil fuels in the future. There is potentially a viable alternative to this dilemma. Its name is hydrogen.
Hydrogen (H) has the Atomic Number of one, meaning it has just one proton in its nucleus, making it the smallest and lightest element in the periodic table. It is also one of the most abundant substances in the entire universe. It does not like to exist as a singular atom, preferring the company of another hydrogen atom and existing as a diatomic gas (H2) that is both colorless and odorless. Hydrogen gas is used commercially in refining petroleum, treating metals, processing fertilizers, and preparing hydrogenated vegetable oils. It found one of its first practical applications as a lifting gas in lighter-than-air aircraft.
The majestic German Zeppelin Hindenburg was filled with hydrogen gas when it flew to America in 1937. While landing at the Lakehurst Naval Air Station in New Jersey, the Hindenburg caught fire and crashed. Its hydrogen lifting gas, somehow lit by a spark, burned ferociously. Hydrogen naturally is prone to mix with oxygen, but not very quickly unless a flame, a spark, or a catalyst is present. In this case, it was believed to be a spark. As a result of the Hindenburg crash, all subsequent lighter-than-aircraft have used helium gas which has slightly less lifting power but is non-flammable. Perhaps the Hindenburg crash images have made people a bit concerned about using hydrogen gas as fuel. Yet many Americans have used gaseous fuels in their homes and our BBQ grilles for years with no problems.

While hydrogen is the most abundant element in the universe, it is not simply found underground where a well or mine can reach it. It is found locked up in other chemical compounds, quite frequently including petroleum. Today, about 95% of the hydrogen we use is produced by reforming methane gas, another fossil fuel. The reformulation, which is done using electricity, produces hydrogen (H2) and carbon dioxide (CO2). The carbon dioxide is then captured and contained to avoid returning it to the atmosphere. If the electricity is produced by burning fossil fuels, the resulting hydrogen would be considered Gray Hydrogen.
The hydrogen would be called Blue Hydrogen, if produced from the same methane feedstock as mentioned before, but the energy used in the reformulation process comes from a renewable source like solar or wind power. Also, the carbon msut be captured for use in other industries.
Green Hydrogen
Finally, if you made hydrogen using electrolysis to separate it from the oxygen in the water, and the energy you used to do so came from renewable sources like wind or solar, it would have the most negligible environmental effect of all. It would be known as Green Hydrogen. Here is where the story gets even more interesting. One of the significant challenges with wind and solar electricity production is matching the demand to energy production. If the sun is bright and the wind is strong, and the load on your grid is not very high, the energy is wasted. If the load is high and the sun is down or the wind is low, the system will crash. You can minimize these problems, if you connect your wind and solar generators directly to an electrolysis system to store the surplus energy in the form of hydrogen gas and oxygen gas. As the sunset or the wind died down, the hydrogen gas generated earlier could be used as fuel to operate a generator to generate electricity. It could be recombined with the oxygen produced earlier in a Fuel Cell to provide additional electricity to meet the load requirements!
If we can scale up the production of Green Hydrogen, the benefits to the environment would be significant. The production processes described above do respond well to being increased in scale. The larger they get, the more efficient they can become. Hydrogen, because it is a gas, can be easily transported by pipeline, stored in pressure vessels, and distributed very much the way we handle natural gas today.
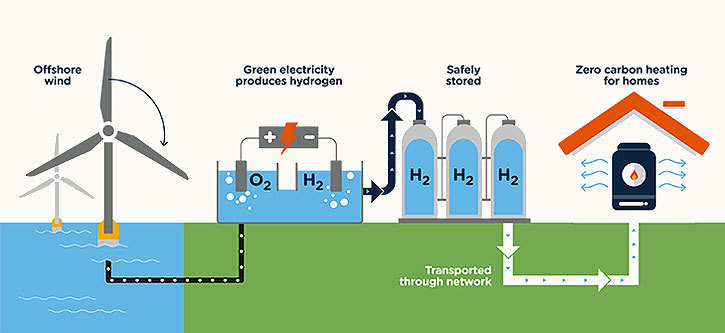
Interestingly, hydrogen can also work as a fuel for internal combustion engines, similar to those found in vehicles today. So, even as new car manufacturers switch to producing electrically powered vehicles, existing internal combustion engine-powered cars will be able to burn Green Hydrogen and produce exhaust gas that is essentially water! Finally, since hydrogen can be recombined with oxygen to generate electricity and water in Fuel Cells, it opens up the world of Fuel Cells' to provide electricity for other applications such as cars, trucks, locomotives, ships, and aircraft or to handle peak load situations on the power grid.
Energy Industry Recognizes The Promise of Green Hydrogen
But what about the petroleum industry that fueled the entire 20th century? They are looking to a green future as well, and in large part, they see it as hydrogen, Green Hydrogen. Saudi Arabia's government plans to make itself the dominant player in what is expected to be a $700 billion market for Green fuels. They have plans to install a gigantic solar farm and an incredible array of wind generators to produce electricity to meet their local needs and create enough Green Hydrogen for exports.

Companies and countries are exploring opportunities in the emerging Green Hydrogen market and the renewable energy economy. This will be, without a doubt, one of the critical areas of growth in the following decades. And it is a dynamic and quickly developing area, so it is worth your attention.
The shift from carbon-based fuels to Green Hydrogen is now clearly on its way and will deliver genuine benefits in sustainability. But as we make the switch, we should all be asking ourselves, is there anything we can do in the meantime to reduce the carbon dioxide in our atmosphere? Here are a few ideas you might consider: reduce your consumption in general, recycle wherever possible, and moderate your use of heating and A/C, turn off gadgets and appliances when you are not using them. You might even consider planting a tree; they "breathe in" carbon dioxide and return oxygen!









